NCATS has recently established the development of A Specialized Platform for Innovative Research Exploration (ASPIRE) to aid in the discovery and development of novel and effective treatments, while at the same time making the process faster and more cost-effective, with a particular focus on pain, OUD and overdose as part of the the Helping to End Addiction Long-term Initiative, or NIH HEAL Initiative. The NCATS ASPIRE Program aims to develop and integrate automated synthetic chemistry, biological screening and artificial intelligence approaches in order to significantly advance our understanding of the relationship between chemical and biological space and enable further access into biologically-relevant chemical space. The ASPIRE platform will utilize currently available knowledge to develop innovative algorithms and predict and synthesize novel structures capable of interacting with specific targets; enable small-scale synthesis of the predicted molecules; and incorporate in-line, rapid biological testing of the molecules. Any new data obtained through this process would then be fed back into the system to further improve design, synthesis and biological characteristics of molecules.
Over 25 million people in the United States experience pain every day (2012 National Health Interview Survey data) and need safe, addiction-free treatments to alleviate their suffering. This clinical demand is of tremendous importance given that overprescribing of opioids for managing acute and chronic pain has fueled the current epidemic of opioid use disorder and overdose deaths, and the effectiveness of opioids for long-term pain management is being questioned. Safe, effective, and non-addictive drugs (small molecules and biologics) to treat pain, mitigate addiction and reverse overdose are key to addressing the opioid crisis. Given failures and limitations of previous drug development efforts, drugs that recognize novel targets, have novel structures, and can be identified in human-based, physiologically relevant in vitro systems are needed. To further advance the NCATS ASPIRE Program and build on the innovations of the ASPIRE Design Challenges, and reward and spur innovative solutions to the development of new drugs for pain, addiction, and overdose, NCATS is issuing this Reduction-to-Practice Challenge to highly collaborative innovators interested in developing working prototypes of novel approaches that would lead to efficacious and non-addictive pain treatments and/or novel treatments for addiction and overdose.
The ultimate goal of the NCATS ASPIRE Program under the HEAL Initiative is the development of a platform that a wide spectrum of scientists can use to advance their translational science relevant to development and pre-clinical testing of new and safer treatments of pain, OUD, and overdose. Further, it is essential that the approach developed in this phase is applicable to any translational problem.
The National Center for Advancing Translational Sciences (NCATS), part of the National Institutes of Health (NIH), is inviting novel solutions for the Reduction-to-Practice Challenge for the NCATS A Specialized Platform for Innovative Research Exploration (ASPIRE) Program. The overall goal of the NCATS ASPIRE Challenges is to reward and spur innovative and catalytic approaches towards solving the opioid crisis through development of: (1) novel chemistries; (2) data-mining and analysis tools and technologies; and (3) biological assays that will revolutionize discovery, development and pre-clinical testing of next generation, safer and non-addictive analgesics to treat pain, as well as new treatments for opioid use disorder (OUD) and overdose. The first phase of these prize competitions was implemented through a suite of concurrent companion design challenges that comprised a separate challenge in each of four areas: chemistry database, electronic laboratory knowledge portal for synthetic chemistry, algorithms, and biological assays; and an additional challenge for a combined solution to at least two challenge areas. For this first phase, innovators submitted designs, not final products or prototypes. Details about the winning submissions to the 2018 ASPIRE Design Challenges can be found at https://ncats.nih.gov/aspire/challenges.
In this second phase, the follow-up reduction-to-practice challenge, the goal is for an open competition to integrate the best designs for a chemistry database, electronic laboratory knowledge portal for synthetic chemistry, algorithms, and biological assays into a single comprehensive platform. Innovators should invoke further scientific and technological development of a comprehensive and integrated solution for the development of new treatments for pain, opioid use disorder and overdose. Innovators design and then demonstrate their integrated solutions and working prototypes, from which winners will be selected.
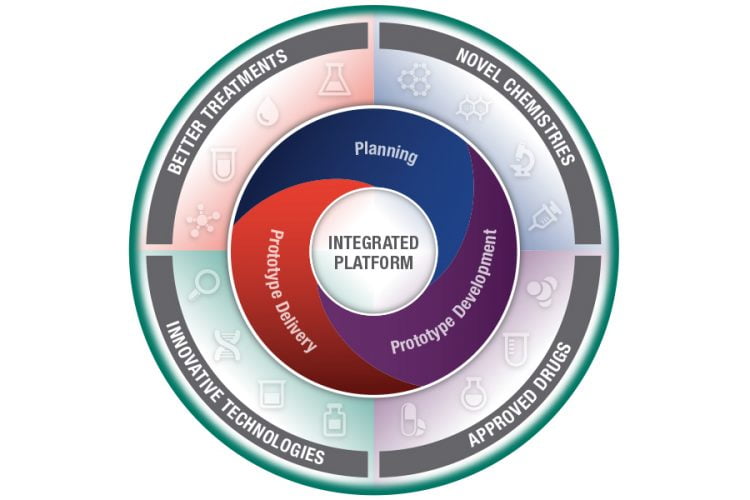
Development of a Comprehensive Integrated Platform for Translational Innovation in Pain, Opioid Use Disorder and Overdose
The goal of this Challenge is to combine the best solutions and develop a working platform that integrates four component areas: Integrated Database, Electronic Synthetic Chemistry Portal, Predictive Algorithms and Biological Assays. The first stage of this Challenge requires submission of a plan for the reduction-to-practice of a platform that integrates the four component areas into a comprehensive solution. The second stage of this Challenge requires the construction and development of a working prototype of this integrated platform. The third stage of this Challenge involves independent testing of the working prototype. In this Challenge, the functionality and degree of integration of the components in the integrated platform/solution will be evaluated. It is anticipated that successful teams for this Challenge will consist of large, multi-disciplinary groups with expertise in all four component areas.
Component Area 1 relates to the development of an open source, controlled access database that incorporates all currently available data of known opioid and non-opioid based analgesics, drugs of abuse, and drugs used to treat drug abuse.
Component Area 2 relates to the development of a open source electronic lab notebook (eLN) that collects, organizes and analyzes data relevant to the chemical synthesis and analyses of known opioid and non-opioid-based analgesics, drugs of abuse and molecules used to treat drug abuse into an electronic laboratory knowledge portal for synthetic chemistry (electronic synthetic chemistry portal; eSCP).
Component Area 3 relates to the development of open source, advanced machine learning algorithms that would facilitate the discovery of novel, efficacious and non-addictive analgesics and/or treatments for drug abuse by utilizing the data collected in open source databases (Component Area 1), eSCPs (Component Area 2), and biological assays (Component Area 4).
Component Area 4 relates to the development of novel, physiologically relevant biological assays that accurately replicate the safety profile and effectiveness of existing drugs to treat addiction and/or overdose, and that can be reliably used in predictive risk assessments of new analgesics or drugs to treat addiction and/or overdose; and/or be able to anticipate the degree of addictiveness of an analgesic prior to clinical testing.
It is anticipated that this Challenge would require large, multi-expert teams. These teams may be assembled from the innovators from the 2018 Design Challenge areas and others who did not participate in the 2018 Design Challenges. All winners of the 2018 ASPIRE Design Challenges are highly encouraged to participate in this ASPIRE Reduction-to-Practice Challenge. Winners of the 2018 ASPIRE Design Challenges may choose to work with other winners from other Challenge areas or to find additional, new collaborators. All submissions, including those from the winners of Challenge 5 from the ASPIRE Design Challenges must propose plans for a solution that integrates all four challenge areas into a comprehensive platform.
Evaluation criteria that judges and technical reviewers will be asked to address are specified below.
Awards:-
The total prize purse is $2,875,000. Prize amounts at each stage for each winning innovator are below. As stated in the Rules for Participating in the Challenge, “innovator” refers to a participating single individual, a participating team, or a participating entity.
Stage 1: Planning: Up to $120,000 per innovator (maximum of 5 innovators). Only the winners of Stage 1 will be invited to participate in Stage 2 of the Challenge.
Stage 2: Prototype Development and Milestones Delivery: Up to four (4) innovators will receive $150,000 upon completion of Milestone 1. Up to three (3) innovators will receive $200,000 for achievement of Milestone 2 and will advance to Stage 3 after completion of Milestone 3. Only the winners of Stage 2 will be invited to participate in Stage 3 of the Challenge.
Stage 3: Prototype Delivery, Independent Validation and Testing: Innovators must successfully complete Milestone 3 before a prototype will be accepted at one or more NCATS-designated laboratories for Stage 3. One (1) grand prize winner will be awarded $750,000, and one (1) runner-up winner will be awarded $325,000.
The NIH reserves the right to cancel, suspend, and/or modify this Challenge at any time through amendment to this notice. In addition, the NIH reserves the right to not award any prizes if no solutions are deemed worthy. The Award Approving Official will be Lawrence A. Tabak, D.D.S., Ph.D., Principal Deputy Director of the National Institutes of Health (NIH).
Payment of the Prize. Prizes awarded under this competition will be paid by electronic funds transfer and may be subject to Federal income taxes. HHS/NIH will comply with the Internal Revenue Service withholding and reporting requirements, where applicable.
Deadline:- 28-02-2021